Efficacy and safety of pilocarpine as a secretagogue versus artificial tears in the management of dry eye disease
Medical hypothesis discovery and innovation in ophthalmology,
Vol. 15 No. 1 (2026),
22 April 2026
,
Page 1-9
Abstract
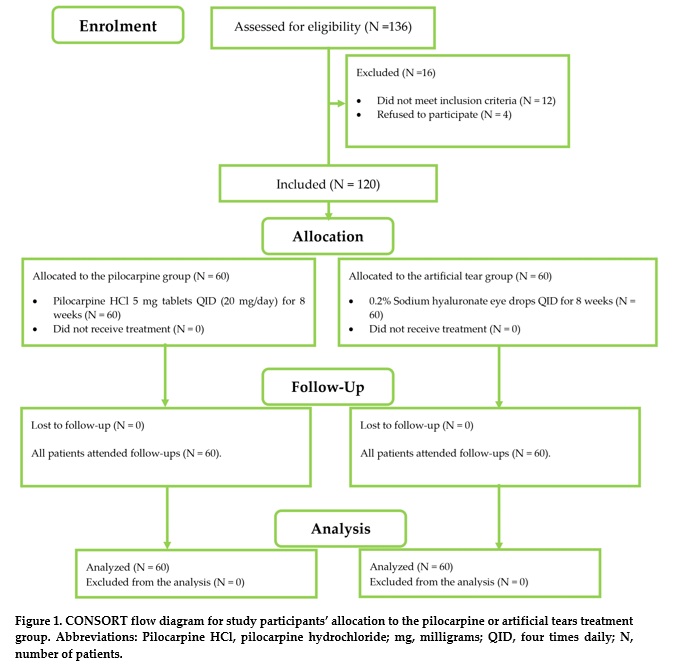
Background: Dry eye disease (DED) is a common multifactorial ocular surface disorder that substantially impairs quality of life and remains among the leading reasons for ophthalmic consultations worldwide. We aimed to compare the efficacy and safety of oral pilocarpine versus artificial tears (AT) in the treatment of DED.Methods: This randomized clinical trial study enrolled patients with DED, randomly allocated to a pilocarpine group receiving 5 mg pilocarpine hydrochloride tablets four times daily (20 mg/day) or an AT group receiving 0.2% sodium hyaluronate eye drops four times daily, for eight weeks. Primary outcomes were changes in Dry Eye Quality of Life Score (DEQS), tear film breakup time (TBUT), and Schirmer’s test after treatment. Secondary outcomes were incidence of adverse events like brow ache, sweating, nausea, headache, diarrhea, and allergic conjunctivitis (AC).
Results: Enrolment comprised 120 patients, randomly assigned to the Pilocarpine group (n = 60) or the AT group (n = 60), with comparable mean age and sex distribution between groups (both P > 0.05). Both groups demonstrated significant post-treatment improvements in DEQS, TBUT, and Schirmer’s test as opposed to baseline (all P < 0.001). The AT group showed a significantly diminished mean (standard deviation [SD]) DEQS (12.1 [2.7] vs. 21.9 [8.4]; P < 0.001) and longer mean (SD) TBUT (11.8 [1.4] s vs. 9.8 [1.8] s; P < 0.05) than the pilocarpine group, while Schirmer’s test results were comparable (P > 0.05). Adverse events were significantly more frequent in the pilocarpine group, with sweating (n = 38, 63%), brow ache (n = 17, 28%), and nausea (n = 15, 25%) occurring exclusively in pilocarpine-treated patients (all P < 0.05); conversely, AC was reported only in the AT group (n = 8, 13%) but did not differ significantly between groups (P > 0.05).
Conclusions: Both pilocarpine and AT produced significant improvements in DED symptoms and objective clinical parameters. However, AT demonstrated superior efficacy in enhancing tear film stability and reducing symptom scores, with a better safety profile. Pilocarpine may still have a role in severe or refractory cases requiring enhanced tear secretion but should be prescribed cautiously due to its systemic cholinergic adverse events.
- Abstract Viewed: 0 times
- Full Text PDF Downloaded: 0 times


