Designing and conducting systematic reviews and meta-analyses in ophthalmology
Medical hypothesis discovery and innovation in ophthalmology,
Vol. 15 No. 1 (2026),
22 April 2026
,
Page 78-89
Abstract
Background: Systematic reviews and meta-analyses represent the highest level of evidence in clinical research, yet their methodological quality in ophthalmology remains inconsistent despite a substantial increase in publication volume. The complexity of existing methodological guidance, such as comprehensive handbooks, may limit their practical use by clinicians and researchers. This study aims to provide a structured, field-specific guide to improve the design and execution of systematic reviews in ophthalmology.Methods: This methodological review synthesizes established international standards and guidelines for systematic reviews, including protocol development, search strategy design, and study selection processes. Key methodological components were critically appraised and adapted to the context of ophthalmic research. The review focuses on the planning and execution phases prior to quantitative synthesis, outlining a stepwise framework encompassing question formulation, preliminary searching, protocol development and registration, database searching, screening, selection, and Risk of Bias (RoB) assessment.
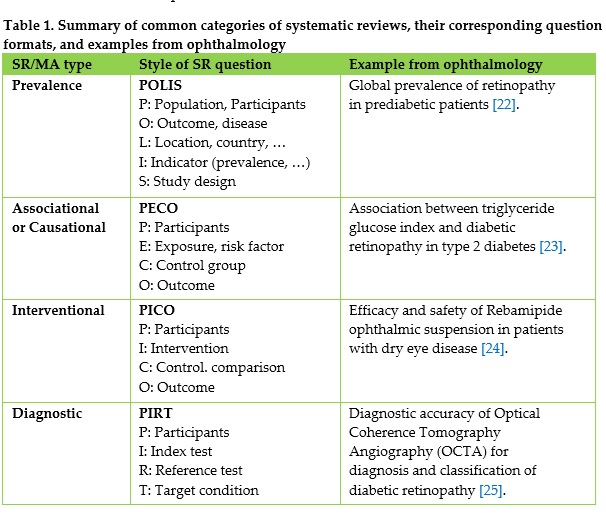
Results: A comprehensive, step-by-step framework for conducting systematic reviews in ophthalmology is presented, structured into four phases and eleven key steps. The review highlights essential methodological considerations, including the formulation of focused research questions using structured frameworks (e.g., PICO and alternatives), the importance of preliminary searches, and the necessity of prospective protocol registration. It emphasizes transparent and reproducible search strategies using multiple databases and gray literature sources, as well as rigorous dual-reviewer screening and selection processes. Additionally, the appropriate application of RoB assessment tools based on study design is detailed. The findings underscore common methodological limitations in ophthalmology reviews, particularly low rates of protocol registration and adherence to reporting standards.
Conclusions: This review provides a practical and structured guide to enhance the methodological quality of systematic reviews in ophthalmology. By translating established methodological standards into a field-specific framework, it aims to improve transparency, reproducibility, and overall research quality. Adoption of these recommendations may address existing gaps in methodological rigor and support the generation of high-quality evidence in ophthalmic research.
- Abstract Viewed: 0 times
- Full Text PDF Downloaded: 0 times


