The Effect of Intravitreal Bevacizumab on Central Serous Chorioretinopathy
Medical hypothesis discovery and innovation in ophthalmology,
Vol. 7 No. 4 (2018),
1 December 2018
,
Page 176-182
Abstract
The aim of this study was to investigate the efficacy of Intravitreal Injection of Bevacizumab (IVB) in patients with Central Serous Chorioretinopathy (CSC) compared to the control group, after four months of injection. In this study, 30 eyes of 30 patients with CSC, who were in the age range of 23 to 50 years old (70% male subject) were included. Eligible patients were randomly allocated to the intervention (n = 15) and control groups (n = 15). Patients in the intervention group received a single dose injection of bevacizumab (1.25 mg in 0.05 mL), while patients in the control group were followed-up during the same time interval, without any medical interventions. Corrected Distance Visual Acuity (CDVA) and Central Macular Thickness (CMT) were evaluated as the primary outcome measures at the four-month follow-up. There was no statistically significant difference between the intervention and control groups regarding their baseline characteristics. Corrected Distance Visual Acuity was improved significantly in the intervention group (P < 0.001), while this improvement was not observed in the control group. Furthermore, greater improvement of CDVA was detected in the IVB group compared to the patients without injection (P = 0.018). The CMT findings were in line with CDVA changes in both groups, revealing a significant reduction of CMT only in the intervention group (P < 0.001). Also, thinner central retina was found in the intervention group compared to the comparison group, at the four-month follow-up (P < 0.001). Based on the findings, bevacizumab could be effective for improvement of both anatomical and functional outcomes in patients with CSC.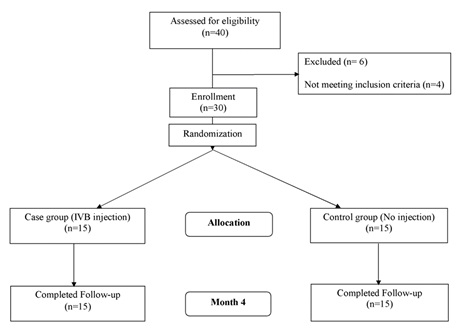
References
Liegl R, Ulbig MW. Central serous chorioretinopathy. Ophthalmologica. 2014;232(2):65-76. doi: 10.1159/000360014 pmid: 24776999
Carvalho-Recchia CA, Yannuzzi LA, Negrao S, Spaide RF, Freund KB, Rodriguez-Coleman H, et al. Corticosteroids and central serous chorioretinopathy. Ophthalmology. 2002;109(10):1834-7. pmid: 12359603
Meyerle CB, Spaide RF. Central serous chorioretinopathy. In: Albert DM, Miller JW, Azar DT, Blodi BA, editors. Albert and Jakobiec's Principles and Practice of Ophthalmology. Philadelphia: Saunders; 2008.
Arora S, Pyare R, Sridharan P, Arora T, Thakar M, Ghosh B. Choroidal thickness evaluation of healthy eyes, central serous chorioretinopathy, and fellow eyes using spectral domain optical coherence tomography in Indian population. Indian J Ophthalmol. 2016;64(10):747-51. doi: 10.4103/0301-4738.194999 pmid: 27905337
Haimovici R, Koh S, Gagnon DR, Lehrfeld T, Wellik S, Central Serous Chorioretinopathy Case-Control Study G. Risk factors for central serous chorioretinopathy: a case-control study. Ophthalmology. 2004;111(2):244-9. doi: 10.1016/j.ophtha. 2003.09.024 pmid: 15019370
Chatziralli I, Kabanarou SA, Parikakis E, Chatzirallis A, Xirou T, Mitropoulos P. Risk Factors for Central Serous Chorioretinopathy: Multivariate Approach in a Case-Control Study. Curr Eye Res. 2017;42(7):1069-73. doi: 10.1080/02713683.2016.1276196 pmid: 28306346
Imamura Y, Fujiwara T, Margolis R, Spaide RF. Enhanced depth imaging optical coherence tomography of the choroid in central serous chorioretinopathy. Retina. 2009;29(10):1469-73. doi: 10.1097/IAE.0b013e3181be0a83 pmid: 19898183
Ross A, Ross AH, Mohamed Q. Review and update of central serous chorioretinopathy. Curr Opin Ophthalmol. 2011;22(3):166-73. doi: 10.1097/ICU.0b0 13e3283459826 pmid: 21427570
Salehi M, Wenick AS, Law HA, Evans JR, Gehlbach P. Interventions for central serous chorioretinopathy: a network meta-analysis. Cochrane Database Syst Rev. 2015(12):CD011841. doi: 10.1002/14651858.CD0118 41.pub2 pmid: 26691378
Ober MD, Yannuzzi LA, Do DV, Spaide RF, Bressler NM, Jampol LM, et al. Photodynamic therapy for focal retinal pigment epithelial leaks secondary to central serous chorioretinopathy. Ophthalmology. 2005;112(12):2088-94. doi: 10.1016/j.ophtha.2005.06 .026 pmid: 16325707
Jung SH, Kim KA, Sohn SW, Yang SJ. Cytokine levels of the aqueous humour in central serous chorioretinopathy. Clin Exp Optom. 2014;97(3):264-9. doi: 10.1111/cxo.12125 pmid: 24417755
Kim M, Lee SC, Lee SJ. Intravitreal ranibizumab for acute central serous chorioretinopathy. Ophthalmologica. 2013;229(3):152-7. doi: 10.1159/00 0345495 pmid: 23257663
Lim JW, Kim MU. The efficacy of intravitreal bevacizumab for idiopathic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2011;249(7):969-74. doi: 10.1007/s00417-010-1581-9 pmid: 21140161
Mehany SA, Shawkat AM, Sayed MF, Mourad KM. Role of Avastin in management of central serous chorioretinopathy. Saudi J Ophthalmol. 2010;24(3):69-75. doi: 10.1016/j.sjopt.2010.03.002 pmid: 23960879
Entezari M, Ramezani A, Yaseri M. Intravitreal bevacizumab for treatment of refractory central serous choroidoretinopathy. Korean J Ophthalmol. 2012;26(2):139-42. doi: 10.3341/kjo.2012.26.2.139 pmid: 22511842
Iacono P, Battaglia Parodi M, Falcomata B, Bandello F. Central Serous Chorioretinopathy Treatments: A Mini Review. Ophthalmic Res. 2015;55(2):76-83. doi: 10.1159/000441502 pmid: 26619293
Artunay O, Yuzbasioglu E, Rasier R, Sengul A, Bahcecioglu H. Intravitreal bevacizumab in treatment of idiopathic persistent central serous chorioretinopathy: a prospective, controlled clinical study. Curr Eye Res. 2010;35(2):91-8. doi: 10.3109/02713680903428306 pmid: 20136418
Aydin E. The efficacy of intravitreal bevacizumab for acute central serous chorioretinopathy. J Ocul Pharmacol Ther. 2013;29(1):10-3. doi: 10.1089/jop.20 12.0072 pmid: 22925113
Chung YR, Seo EJ, Lew HM, Lee KH. Lack of positive effect of intravitreal bevacizumab in central serous chorioretinopathy: meta-analysis and review. Eye (Lond). 2013;27(12):1339-46. doi: 10.1038/eye.2013 .236 pmid: 24202051
Park SU, Lee SJ, Kim M. Intravitreal anti-vascular endothelial growth factor versus observation in acute central serous chorioretinopathy: one-year results. Korean J Ophthalmol. 2014;28(4):306-13. doi: 10.3341/kjo.2014.28.4.306 pmid: 25120339
Guyer DR, Yannuzzi LA, Slakter JS, Sorenson JA, Ho A, Orlock D. Digital indocyanine green videoangiography of central serous chorioretinopathy. Arch Ophthalmol. 1994;112(8):1057-62. pmid: 8053819
Pryds A, Sander B, Larsen M. Characterization of subretinal fluid leakage in central serous chorioretinopathy. Invest Ophthalmol Vis Sci. 2010;51(11):5853-7. doi: 10.1167/iovs.09-4830 pmid: 20505211
Alkin Z, Osmanbasoglu OA, Ozkaya A, Karatas G, Yazici AT. Topical nepafenac in treatment of acute central serous chorioretinopathy. Med Hypothesis Discov Innov Ophthalmol. 2013;2(4):96-101. pmid: 24822228
Chan WM, Lam DS, Lai TY, Tam BS, Liu DT, Chan CK. Choroidal vascular remodelling in central serous chorioretinopathy after indocyanine green guided photodynamic therapy with verteporfin: a novel treatment at the primary disease level. Br J Ophthalmol. 2003;87(12):1453-8. pmid: 14660450
Tzekov R, Lin T, Zhang KM, Jackson B, Oyejide A, Orilla W, et al. Ocular changes after photodynamic therapy. Invest Ophthalmol Vis Sci. 2006;47(1):377-85. doi: 10.1167/iovs.05-0838 pmid: 16384988
Lee JY, Chae JB, Yang SJ, Kim JG, Yoon YH. Intravitreal bevacizumab versus the conventional protocol of photodynamic therapy for treatment of chronic central serous chorioretinopathy. Acta Ophthalmol. 2011;89(3):e293-4. doi: 10.1111/j.1755-3768.2009.01 835.x pmid: 20346078
Robertson DM, Ilstrup D. Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am J Ophthalmol. 1983;95(4):457-66. pmid: 6682293
Koss MJ, Beger I, Koch FH. Subthreshold diode laser micropulse photocoagulation versus intravitreal injections of bevacizumab in the treatment of central serous chorioretinopathy. Eye (Lond). 2012;26(2):307-14. doi: 10.1038/eye.2011.282 pmid: 22079961
Lim JW, Kim MU, Shin MC. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina. 2010;30(9):1465-71. doi: 10.1097/IAE.0b013e3181d8e7fe pmid: 20526231
Torres-Soriano ME, Garcia-Aguirre G, Kon-Jara V, Ustariz-Gonzales O, Abraham-Marin M, Ober MD, et al. A pilot study of intravitreal bevacizumab for the treatment of central serous chorioretinopathy (case reports). Graefes Arch Clin Exp Ophthalmol. 2008;246(9):1235-9. doi: 10.1007/s00417-008-0856-x pmid: 18523796
Gregori-Gisbert I, Aguirre-Balsalobre F, Garcia-Sanchez J, Leon-Salvatierra G, Mengual-Verdu E, Hueso-Abancens JR. [Recurrent and chronic central serous chorioretinopathy. Retina thickness evaluation one month after intravitreal bevacizumab injection]. Arch Soc Esp Oftalmol. 2011;86(12):407-11. doi: 10.1016/j.oftal.2011.05.021 pmid: 22117740
Lim JW, Ryu SJ, Shin MC. The effect of intravitreal bevacizumab in patients with acute central serous chorioretinopathy. Korean J Ophthalmol. 2010;24(3):155-8. doi: 10.3341/kjo.2010.24.3.155 pmid: 20532141
Akhlaghi MR, Nasrollahi C, Namgar SM, Kianersi F, Dehghani AR, Arefpour R. Effect of Intravitreal Injection of Bevacizumab on Acute Central Serous Chorioretinopathy Patients Who Visited Feiz Hospital during 2014-2015 Period. Adv Biomed Res. 2017;6:125. doi: 10.4103/abr.abr_193_15 pmid: 29142888
Semeraro F, Romano MR, Danzi P, Morescalchi F, Costagliola C. Intravitreal bevacizumab versus low-fluence photodynamic therapy for treatment of chronic central serous chorioretinopathy. Jpn J Ophthalmol. 2012;56(6):608-12. doi: 10.1007/s10384-012-0162-3 pmid: 22915299
Gass JD. Pathogenesis of disciform detachment of the neuroepithelium. Am J Ophthalmol. 1967;63(3):Suppl:1-139. pmid: 6019308
- Abstract Viewed: 1502 times
- Full Text PDF Downloaded: 1093 times


